 
It is incredibly reactive and is never found in its elemental state in nature. Chlorine has a high electronegativity, only behind oxygen and its close cousin, fluorine. It is a green gas at room temperature, and its name comes from the Greek word chloros, meaning 'green'. 
In fact, polytetrafluoroethylene has the third-lowest friction coefficient of any known solid, and is the only material a gecko can’t stick to! ChlorineĬhlorine, with an atomic number of 17, is the next smallest member of the halogens group. It is also extremely slippery, which is why it is often used in non-stick pans. C-C and C-F bonds are extremely strong, which means that the polymer doesn’t react with much else. Teflon™ is a brand name for the compound polytetrafluoroethylene, a polymer made from chains of carbon and fluorine atoms. Nowadays fluorine is added to toothpaste and is a part of Teflon™.įor more on CFCs, check out Ozone Depletion. In the 1900s it was used in refrigerators in the form of CFCs, or chlorofluorocarbons, which are now banned due to their harmful effect on the ozone layer. It was originally used to lower the melting points of metals for smelting. The name fluorine comes from the Latin verb fluo-, meaning 'to flow', which reflects its origins. It is stored in special containers using metals such as copper, which form a protective layer of fluoride on their surface. Fluorine reacts readily with almost all other elements and can even react with glass. It is the most electronegative element in the periodic table, making it one of the most reactive elements. Fluorineįluorine is the smallest and lightest member of the group, with an atomic number of 9 and a pale yellow gas at room temperature. Let’s now take a look at the elements individually, starting with fluorine. The sixth is the artificial element tennessine, and you’ll find out why some people don’t include it in the group later on. The fifth member is astatine, an extremely radioactive element. These are fluorine, chlorine, bromine, and iodine. The first four members are known as the stable halogens. The elements in the halogensĪt the start of this article, we said that the halogen group contains six elements. If you would like to see halogen reactivity in action, visit Reactions of Halogens. You'll learn more about these trends in Properties of Halogens. Oxidising ability decreases going down the group whilst reducing ability increases. Reactivity and electronegativity decrease going down the group whilst atomic radius and melting and boiling points increase. And finally, halogens form diatomic molecules - that means they're usually found in pairs of two.Ī chlorine atom, left, and chloride ion, right The first four halogens typically form anions with a charge of -1, meaning they've gained one electron. Halogens also form anions, which are negatively charged ions. In fact, fluorine has the highest electronegativity value of any element on the periodic table. 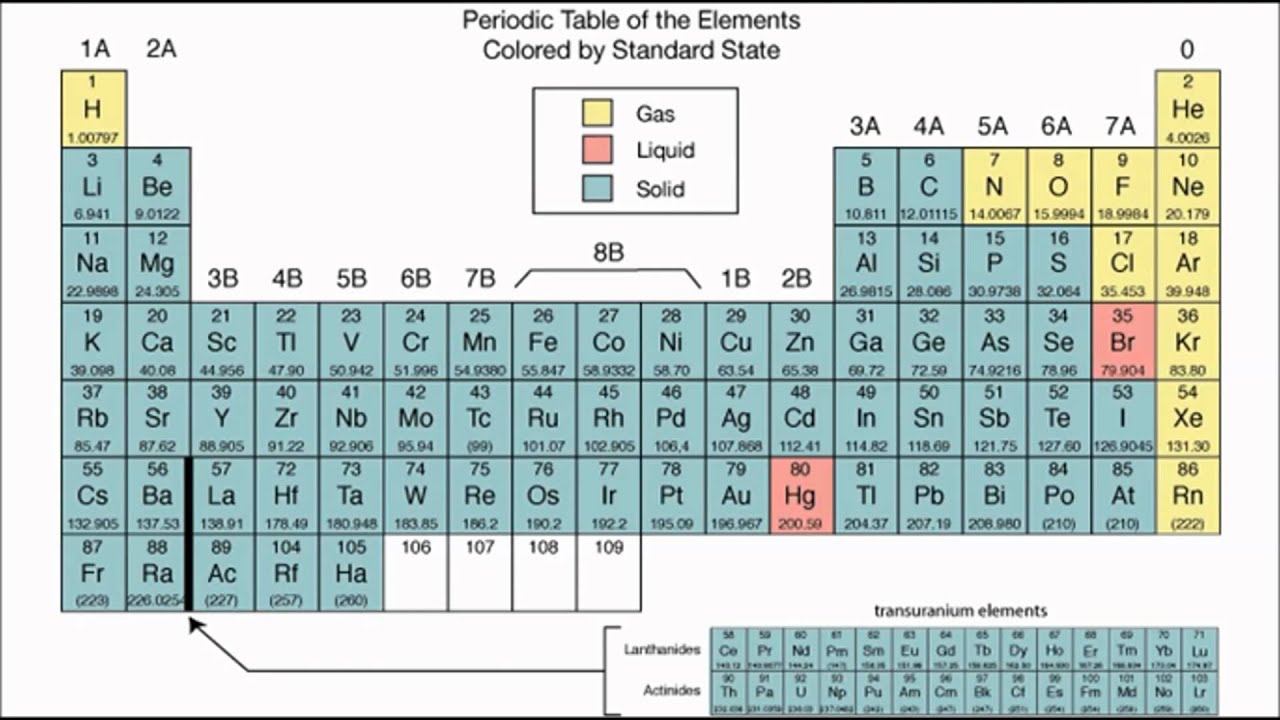
Another cool thing about halogens is that they have high electronegativity values. 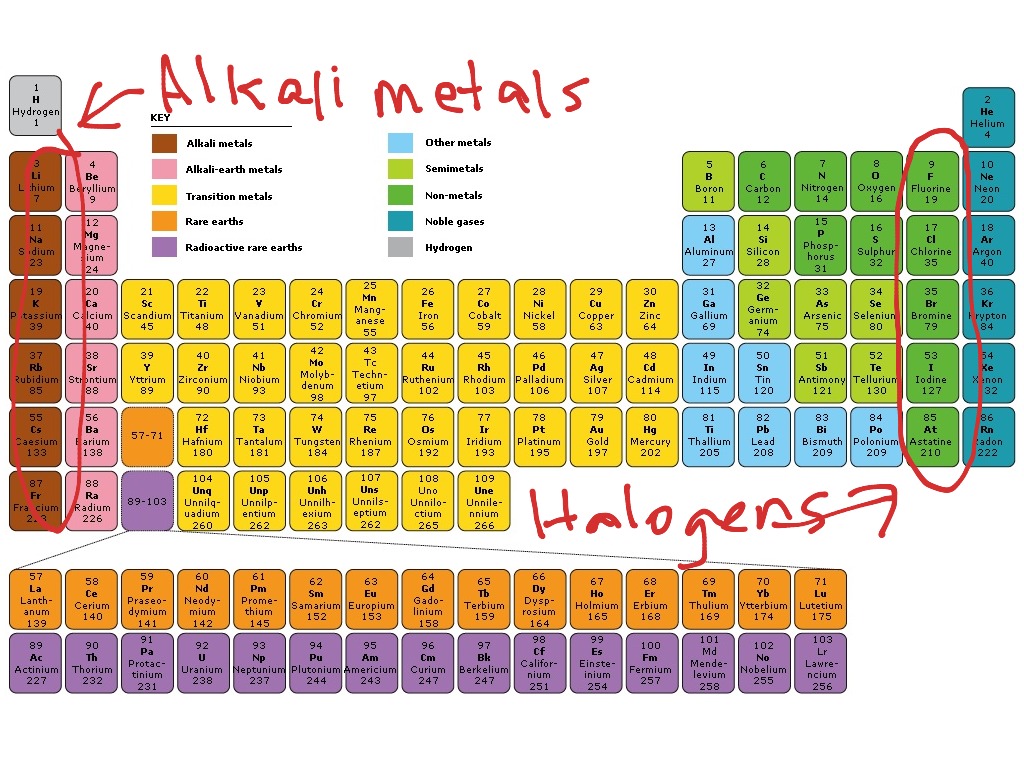
They also have low melting and boiling points. Oh, and here's something interesting - they can sublime easily, meaning they can go straight from a solid to a gas without becoming a liquid first. Plus, when they're in their solid form, they're dull and brittle. They're not great at conducting heat or electricity, and they produce acidic oxides. Properties of halogensĭid you know that the halogens are actually non-metals? That means they have a lot of similar properties to other non-metals you might know about. Finally, we’ll also explore how you can test for the presence of halide ions in compounds. We’ll then outline some of the reactions they take part in and their uses. We’ll look at their properties and characteristics before taking a closer look at each member in turn. This article is an introduction to the halogens. Group 7 or group 17? Sometimes it is just easier to refer to them as 'the halogens' 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
